We know that compounds can be acidic, basic, and neutral. Looking at pKa and functional groups can help us further in selecting the right SPE sorbent and better understanding how it works with our analyte of interest. Let’s take a look at the compound dopamine:
Figure 1. Structure of dopamine.
When we look at the structure, we can see the conjugation through the benzene ring. This conjugation will increase our hydrophobic bonding. Now let’s look at the pKa of the functional groups.
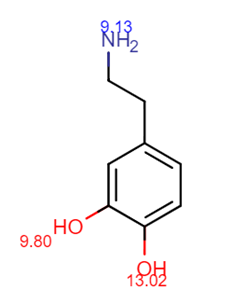
Figure 2. Dopamine pKas are added per Chemicalize.
In Figure 2, we can see that our most acidic alcohol functional group has a pKa of 9.80 and our strongest amine basic functional group has a pKa of 9.13. Remembering some fundamentals of general chemistry, we know strong acids and strong bases are ionized over pretty much the entire usable pH range. Well, the opposite is true for weak acids and weak bases. Ionization for weak acids and bases can be controlled (effectively turned on or off) by manipulation of pH. So what constitutes strong vs weak? Let’s take a look at the sorbent chemistry selection guide in the EVOLUTE® Express User Guide.
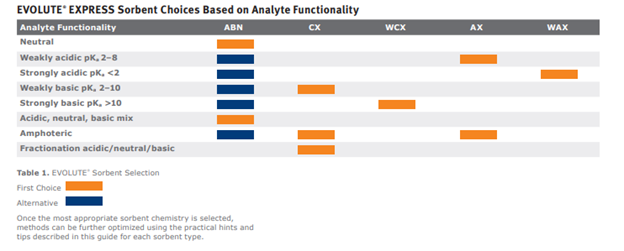 Figure 3. Looking at the analyte functionality column we can see the pKa ranges for each categorization, we will use this to help us determine the best sorbent chemistry for analysis.
Figure 3. Looking at the analyte functionality column we can see the pKa ranges for each categorization, we will use this to help us determine the best sorbent chemistry for analysis.
The pKas for the acidic functional groups fall past the weakly acidic analyte functionality at 9.80 and 13.02. So we know these aren’t anything we can exploit. Looking at the strongest basic functional group pKa at 9.13, this falls within our “weakly basic” category. This means it will hold a positive charge in a neutral or acidic pH environment. For a quick refresher on pH adjustment refer to the pH adjustment sample prep blog. Since we have a weakly basic functional group, we will exploit that ion exchange potential by using the EVOLUTE®CX sorbent.
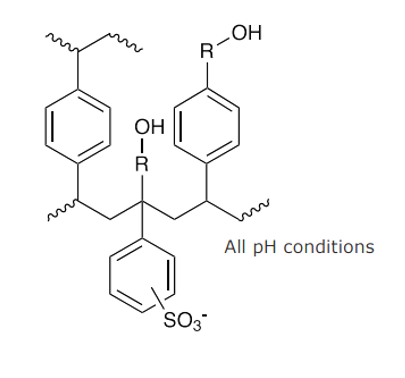
Figure 4. EVOLUTE Express CX Sorbent, notice the modified polysystrenedivinylbenzene polymer which allows the sorbent to be water-wettable.
The CX sorbent has non-polar (hydrophobic) and strong cation exchange functionality. There is a sulfonic acid functional group that is negatively charged to retain positively charged analytes. We can see in Figure 4 the hydrophobic and ionic interactions. The acidic functional group has a pKa of ~-7 which tells us it is going to be 100% ionized over the entire pH range. We can also see the reverse phase interaction portion of the structure that will allow for hydrophobic interactions.
Figure 5. Here we see where the hydrophobic interactions are occurring between the CX sorbent and the charged dopamine molecule during loading. We also see the ionic interactions that are simultaneously occurring, thereby creating the mixed mode interactions.
Now let’s see how each step occurs in the CX sorbent as seen in Current Methodologies for Drugs of Abuse Urine Testing.
In this example, we are working with human serum and will use the EVOLUTE®EXPRESS CX 30 mg 96- well plate part number: 601-0030-PX01.
| Step | Description | What’s happening? |
| Sample Pre-treatment | 100 µL 4% H3PO4 |
Diluting the sample will help decrease viscosity allowing it to more readily flow through the SPE sorbent. Remembering the pH rule we will pre-treat the sample to 2 units lower than the pKa to ensure 100% ionization of our analyte. The addition of phosphoric acid will ensure maximum ionization of our basic functional groups. |
| Condition (optional) | 1 mL methanol | This is optional when using the EVOLUTE Express line due to the water-wettable functional groups and frits. |
| Positive Pressure |
Recommend positive pressure. 1 min, 2 bar |
Positive pressure avoids variable well-to-well flow-through. Nitrogen is typically preferred to prevent oxidation and purity prevents contamination. |
| Equilibration (optional) | 1 mL 4% H3PO4 |
This is optional when using the EVOLUTE Express line due to the water-wettable functional groups and frits. |
| Positive Pressure |
Recommend positive pressure. 1 min 2 bar |
Positive pressure avoids variable well-to-well flow-through. Nitrogen is typically preferred to prevent oxidation and purity prevents contamination. |
| Sample Loading | 200 µL pretreated sample | This is within the recommended load capacity. |
| Positive Pressure |
Recommend positive pressure. 1 min, 0.5 bar |
Positive pressure avoids variable well-to-well flow-through. Nitrogen is typically preferred to prevent oxidation and purity prevents contamination. |
|
Wash 1 |
1000 µL 4% H3PO4 |
Maintains maximum ionization of our basic functional groups. Remove polar interferences such as salts and small proteins. |
| Positive Pressure |
Recommend positive pressure. 1 min, 0.8 bar |
Positive pressure avoids variable well-to-well flow-through. Nitrogen is typically preferred to prevent oxidation and purity prevents contamination. |
| Wash 2 | 1 mL 50:50 methanol/water |
Removes hydrophobic interactions, simultaneously releasing hydrophobic compounds such as phospholipids and lysophospholipids. |
| Positive Pressure |
Recommend positive pressure. 1 min, 0.8 bar |
Positive pressure avoids variable well-to-well flow-through. Nitrogen is typically preferred to prevent oxidation and purity prevents contamination. |
| Elution | 2 x 750 µL 78:20:2 dichloromethane/ methanol/ and ammonium hydroxide |
Adding the basic buffer will neutralize the analyte breaking the ionic interactions, thereby releasing the analytes from the sorbent. The organic solvents will break any remaining hydrophobic interactions with the analyte. |
| Positive Pressure |
Recommend positive pressure. 1 min, 0. Bar |
Positive pressure avoids variable well-to-well flow-through. Nitrogen is typically preferred to prevent oxidation and purity prevents contamination. |
| Dry Down | Stream of air or nitrogen | Nitrogen is typically preferred to prevent oxidation and purity prevents contamination. |
| Reconstitution | 90:10 0.1 formic acid in water/ 0.1% formic acid in methanol | Mass spectrometry suitable solvent. |
The typical process for CX sample preparation is based on both reverse phase/ hydrophobic interactions and ion-exchange abilities of your analytes of interest. In figure 4 we mentioned the added benefit of the polymeric sorbent. Why is this important? In traditional silica-based SPE, you have to condition and equilibrate your sorbent and frits, which were hydrophobic. With this polymeric SPE product, both the sorbent and frits are water-wettable and with an aqueous sample, the sorbent is able to interact with your sample without conditioning and equilibration. This also has the benefit of removing the variability of wells drying out during your analysis. This is especially beneficial if you are working with a 96-well plate and well A1 might be solvated several minutes before H12; the water-wettable capability of the EVOLUTE Express line minimizes the risk of wells drying and well-to-well variability in your application results.

