Cl-trityl resin can be tricky to use not only from an amino acid loading perspective, but one must also consider the highly labile linkage to the solid support1. This lability is advantageous in instances where fully protected peptides are required for subsequent off-resin chemistry. However, due to the mild acidic conditions necessary for cleavage, concern has arisen that the use of particular coupling reagents, especially at elevated temperatures, may be enough to liberate peptide from the resin. This premature cleavage leads to mass loss, requiring additional syntheses to meet required product yields. Herein I explore whether the DIC and Oxyma coupling reagent system does cause crude mass loss and evaluate strategies to ensure that recovery is maximized.
As I mentioned, the DIC/Oxyma coupling system is acidic and can cause peptides to be cleaved over the course of a synthesis, especially for longer sequences2. To combat this, some labs have used base to change the pH of the system thereby reducing the potential for premature cleavage. I decided to use DIEA as the base because it is commonly used in Fmoc-SPPS and is not strong enough to remove Fmoc protecting groups. I checked three different conditions: 0, 0.4, and 1.0 equivalents of DIEA using our standard coupling conditions on a Biotage® Initiator+ Alstra™ peptide synthesizer. I used ~200 mg of glycine-loaded Cl-trityl ChemMatrix® resin for each experiment and measured the recovered crude mass for each experimental condition. The peptide I chose was ACP however, I forgot that I was using preloaded resin, so, the sequence synthesized was ACP+G (VQAAIDYINGG).
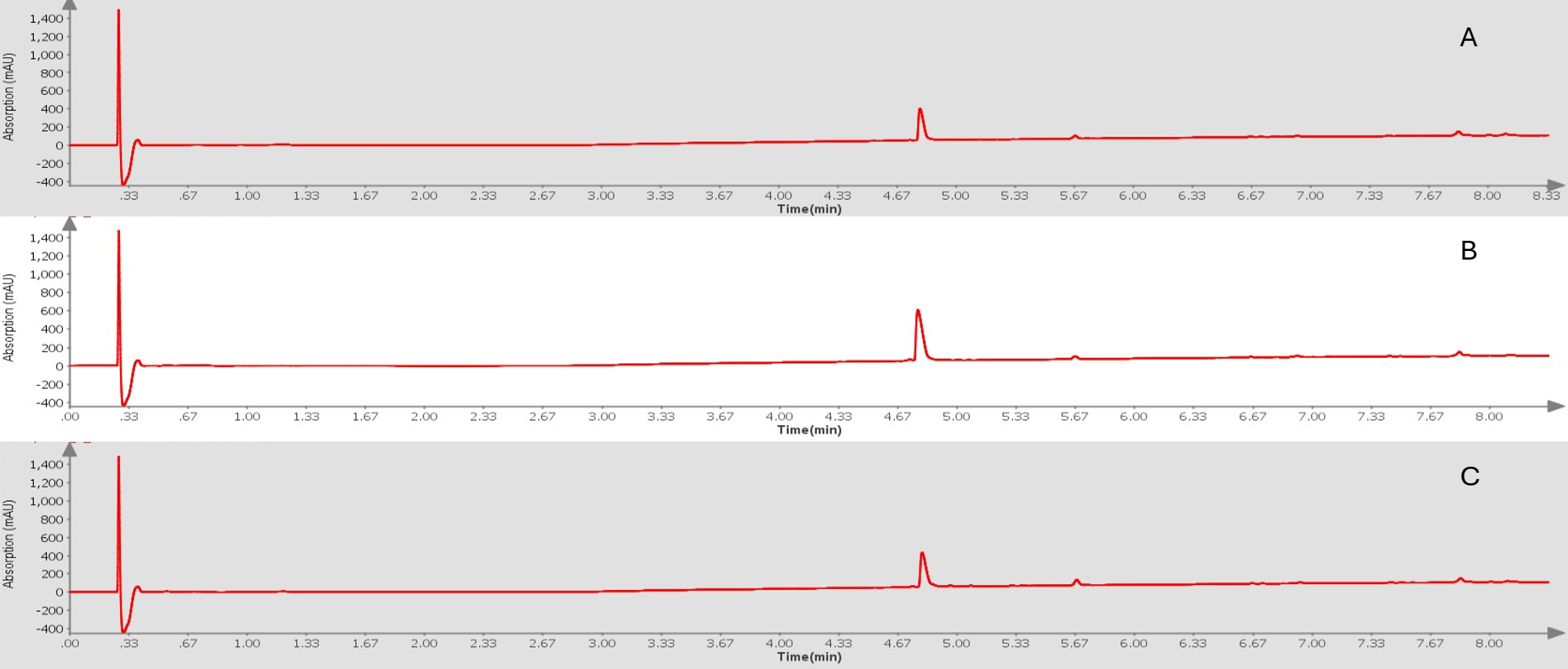
Figure 1. Analytical HPLC chromatograms for crude ACP+G peptides synthesized using 0 eq. of DIEA (A), 0.4 eq. of DIEA (B) or 1 eq. of DIEA (C). Similar purity shows DIEA does not disrupt DIC/Oxyma coupling efficiency. Peptide identity confirmed by mass spec, data not shown.
As you can see in Figure 1, the syntheses were all successful from a crude purity perspective. This allows us to more readily correlate the recovered crude mass to the impact of increasing concentrations of base in the coupling reaction mixture. Table 1 depicts the recovered crude mass for all conditions evaluated. Not surprisingly, the 0 eq synthesis had less collected mass than both the 0.4 eq and 1.0 eq DIEA syntheses even given the same starting resin amount.
| Table 1. Recovered Mass of Crude Peptides | |
| Eq. of DIEA | Collected Mass (mg) |
| 0.0 | 24.9 |
| 0.4 | 42.9 |
| 1.0 | 33.5 |
All in all, this data suggests that adding DIEA will likely increase peptide yield when using DIC/Oxyma coupling conditions at elevated temperatures. Increasing the amount of DIEA per coupling does seem to have a negative impact on yield but this may be due to other contributing factors. For example, my resin was 2 weeks older for my final reaction and could have contained higher moisture content compared to the other 2 syntheses inflating weighed resin mass- I noticed that it was visibly stickier compared to the 0eq and 0.4 eq reactions. Higher DIEA could reduce coupling efficiency but for that I would have expected a decrease in crude purity. I also notice during the washing and swelling steps of the final reaction I had a significant large clump of resin that didn't seem to break up even under mixing conditions. With this there could be less access to the resin and therefore less active sites would be available for loading and synthesis.
If you want to learn more about peptide synthesis techniques check out our other blogs or click the link below to learn more about my peptide workflow!
Learn More
1) C. Echalier, Amino Acids, 2013, 45, 1395-1403
2) S. Manne, ACS Omega, 2022, 7, 6007-6023

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership