While polar compound purification is frequently performed using a silica column and a DCM/MeOH solvent system, there is a need to reduce its use in chromatography for a number of reasons. First, dichloromethane (DCM) is categorized as a health hazard (neurotoxin[1]) and likely carcinogen (per the US EPA). If that isn’t a good enough reason, DCM is not environmentally friendly carrying a large carbon footprint and being a greenhouse gas[2].
Now that everybody is on-board with why DCM should not be used in flash chromatography, what are the alternative purification strategies? Well, you can always try a crystallization or trituration strategy, which could take a while to optimize or, you can go to an alternative flash chromatography strategy to quickly and effectively get the job done.
Besides DCM’s toxicity and sustainability issues, chemists are often concerned about silica solubility in methanol as well as a flash system’s ability to accurately create a shallow gradient. I can assure you that with today’s spherical silica packed flash columns the methanol-silica dissolution issue has been eliminated. The gradient accuracy situation, though, will vary between system manufacturers. In my 40+ years of chromatography experience, however, I have learned that if a shallow gradient is needed, chances are it is not the optimal strategy for that sample’s separation or purification.
Alternatives to DCM/MeOH have been researched and published including the use of (3:1 ethyl acetate/isopropanol) with hexane or heptane (or other variations on that theme). While generally effective (I use this solvent system when needed for some of my research), these alternative solvents are far from being green. Instead, I have found reversed-phase flash chromatography very efficient and effective for both purification and sustainability.
Take, for example, the purification of a reaction I performed in my lab (nicotinuric acid with benzylamine). TLC with normal-phase solvents such as DCM/MeOH and EtOAc/IPA showed the DCM solvent system was the better choice, unfortunately. When I performed the purification (59 mg load on a 10-gram Biotage® Sfär HC column) using this methodology, it initially appeared to generate a good purification, Figure 1.
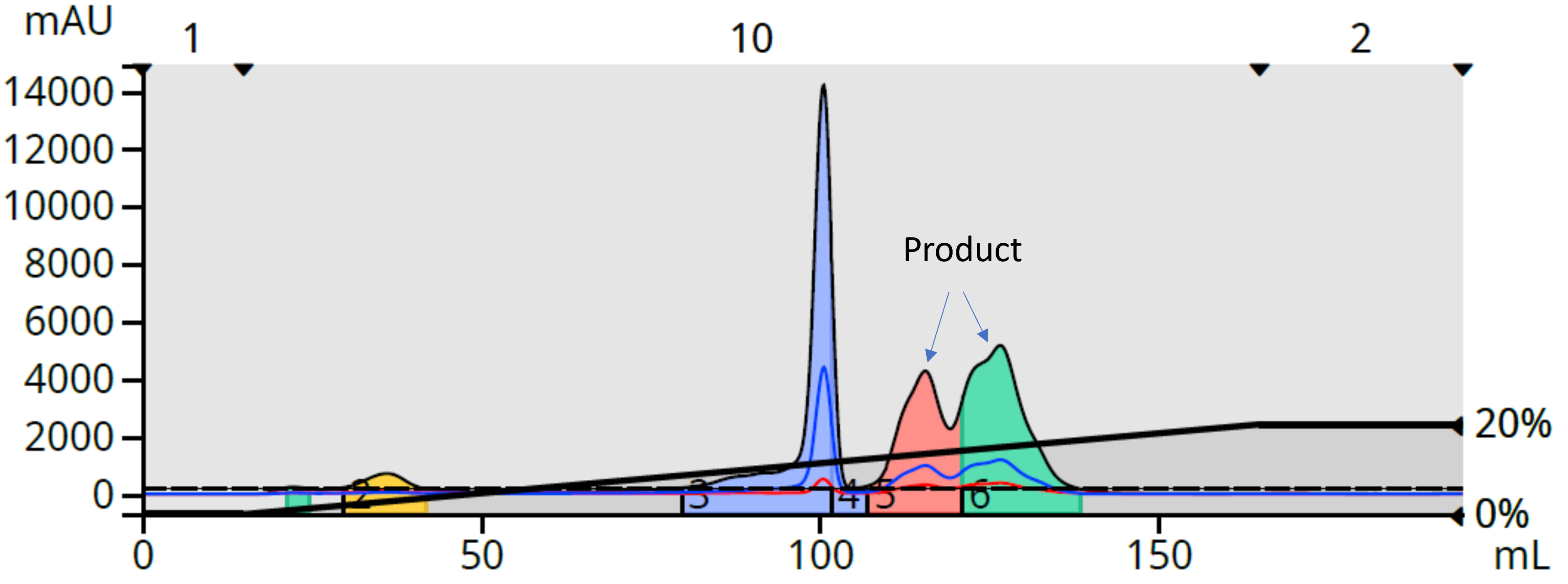
Figure 1. DCM/MeOH (0-20%) reaction mixture gradient purification separated the more lipophilic by product from the two product peaks which also have partially separated.
Upon further inspection and fraction analysis, although a complete separation was achieved between the product and its largest by-product, the product itself eluted in two peaks and both fractions were highly colored, Figure 2. These were not isomers but the same compound likely exhibiting solubility issues in the mobile phase (DCM) and contaminated with something yellow. That’s another potential problem with the DCM/MeOH purification approach – potential insufficient solubility in the mobile phase.

Figure 2. Picture of the dried DCM/MeOH product fractions showing their contamination.
As a greener alternative to the DCM/MeOH purification, I tried reversed-phase with a 12-gram Biotage® Sfär C18 column. What I found was that reversed-phase flash worked very well for this purification using water and acetonitrile (methanol also would work), Figure 3.
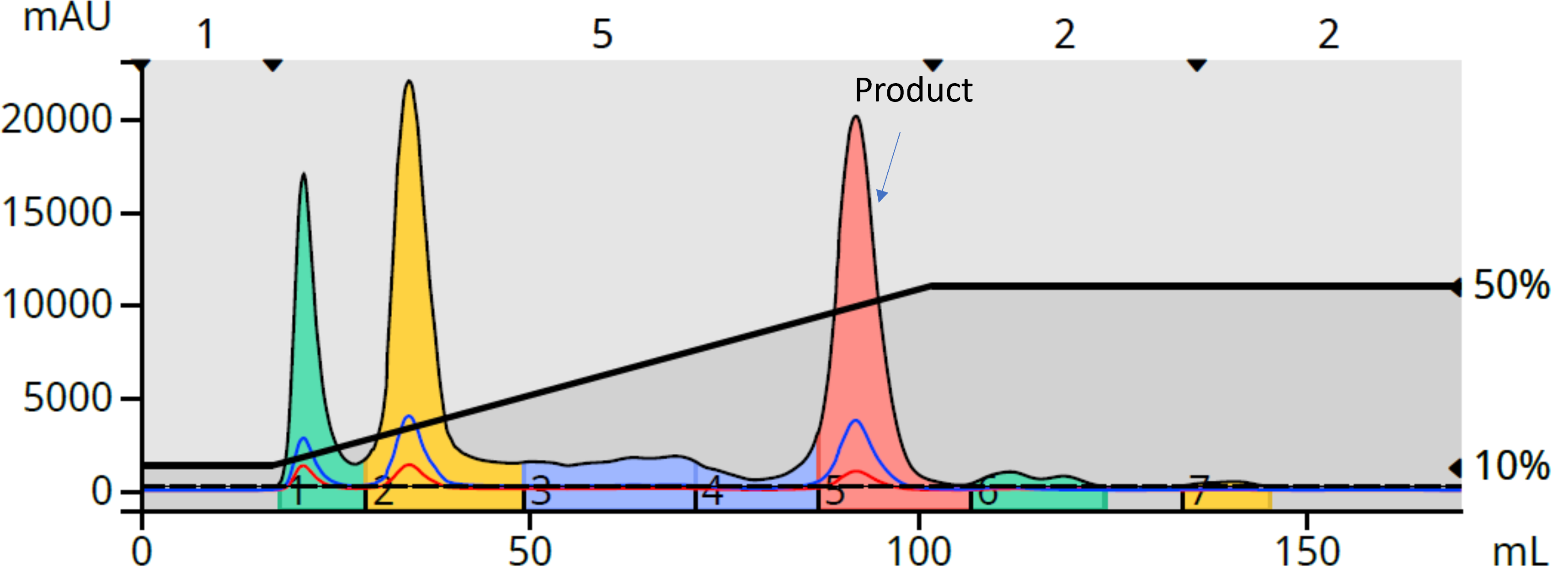
Figure 3. Reversed-phase flash purification of the reaction mixture shows a more complete separation of the target compound from the major by-products and unreacted starting materials.
The reversed-phase method retained the reaction product very well while allowing the more polar by-products and unreacted starting materials to elute quickly. Also important to note is the reversed-phase method used ~13% less solvent (170 mL vs. 195 mL), contained more target compound (27 mg) than what was recovered using the normal-phase, DCM-MeOH purification (21.7 mg), and its product fraction was colorless (dried as white residue), Figure 4.

Figure 4. Photo of the dried, white, reversed-phase product fraction.
As they say in tennis – game-set-match!
Clearly, reversed-phase flash chromatography should be your go-to purification strategy, even with the need to evaporate water. It’s a better, more efficient, and more sustainable strategy, especially since the reversed-phase columns are re-usable!
Looking for more tips on greener purification? Visit our website and download our webinar, Green Flash Chromatography - Idealistic, Realistic, Practical, Attainable?
[1] Vedantu.com
[2] MedChem Tips and Tricks - American Chemical Society (acs.org)


