Jan 30, 2023 5:39:31 PM
Peptide purification with flash column chromatography - a beginner's experience
By Elizabeth Denton

As a peptide chemist, I was trained to purify my peptides with reversed-phase HPLC, just as many a peptide chemist before me. Despite the hundreds of hours I’ve logged in front of an HPLC, injecting samples and collecting peak fractions, I can’t imagine using any other method to purify my freshly synthesized and cleaved peptides. In fact, you’d be hard pressed to convince me to try something else. But here I am, trying something new. Wish me luck!
In this post, I’ll describe my experiences using flash chromatography to purify a new peptide sample.
Flash column chromatography? Why is this better than what I am currently doing for peptide purification? What do you mean I can inject my ENTIRE synthesis onto the column? It will still get pure? Really? How much of my hard-earned peptide (yes, I have had the pleasure of manual solid phase peptide synthesis) is going to get stuck on all this silica? How will my tried-and-true chromatography gradients translate to a flash system? Can I reuse the cartridges like my HPLC columns? You mean I have to learn ANOTHER set of software too? Bonus, can I use mass-directed purification to increase my first-pass purity?
Thankfully, my lab is equipped with a Biotage IsoleraTM that has AssistTM software. Think of Assist as the easy button road map through the software for users that have very little experience with flash chromatography. Orange buttons direct you through each window of the method setup and generates a basic chromatography method without much user intervention. It even tells me which cartridge to use!
I’ve had some generic peptide laying around for a while, so I decided to purify the sample and approach this with a completely fresh perspective. With little previous chromatographic knowledge regarding this specific peptide, I let the system define the mobile phase gradient. The default gradient holds at 10 % acetonitrile for 1 column volume (CV), then increases acetonitrile concentration from 10 % to 100 % over 10 CVs and finally washes the column at 100 % acetonitrile for an additional 2 CVs. The entire method requires less than 25 minutes and only 400 mL of mobile phase! I simply selected the smallest C18 cartridge from the options presented by the software, a 12 gram Biotage® SNAP-KP-C18 (275 mg capacity), primed the cartridge, dissolved my peptide in DMSO, injected approximately 20 mg of peptide and pushed play, Figure 1.
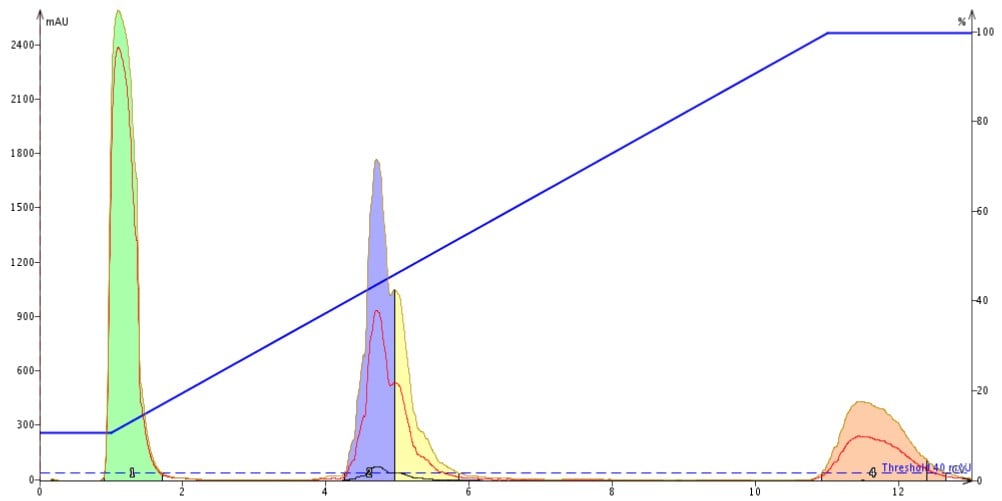 Figure 1: The default reversed-phase gradient increases acetonitrile concentration from 10% to 100% over 10 column volumes (12 minutes) correlating to a gradient of approximately 7.5% per minute.
Figure 1: The default reversed-phase gradient increases acetonitrile concentration from 10% to 100% over 10 column volumes (12 minutes) correlating to a gradient of approximately 7.5% per minute.
Well… ok. Luckily the SNAP-KP-C18 columns are reusable because that doesn’t look like very good separation at all! Full disclosure, I didn’t expect the cleanest of chromatograms after storing the peptide in solution for several weeks prior to this purification. I did learn though, approximately what concentration of acetonitrile is required to elute my product(s). In order to rework my gradient and hopefully improve the purification, I simply moved each of the endpoints to the desired acetonitrile concentration while maintaining the gradient duration. The new gradient more closely resembles a standard HPLC gradient and it was much easier than modifying my previous HPLC methods! Upon initial inspection the separation improved, revealing several other peptide products that I anticipated would exist. The resolution of each peak however, leaves something to be desired, Figure 2.
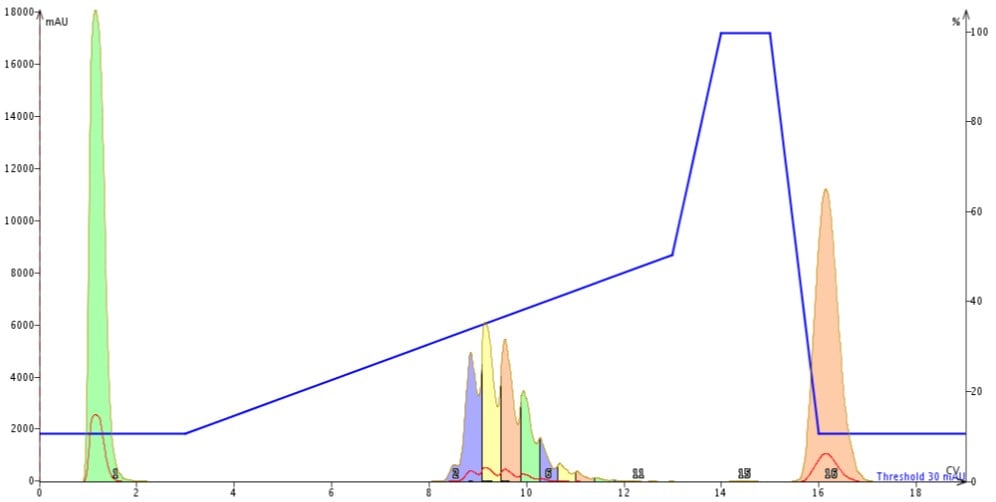 Figure 2: Results of revised mobile phase gradient in which acetonitrile concentrations increase from 10% to 50% over 10 column volumes followed by a short column wash and reequilibration.
Figure 2: Results of revised mobile phase gradient in which acetonitrile concentrations increase from 10% to 50% over 10 column volumes followed by a short column wash and reequilibration.But then I thought about gradient optimization in standard RP-HPLC. So I next extended the gradient an additional 5 CVs. As I expected, this continued to improve my peak separations, Figure 3. So far so good! My standard HPLC separation optimization strategies still seem to work and we’ll save gradient optimization for another discussion later.
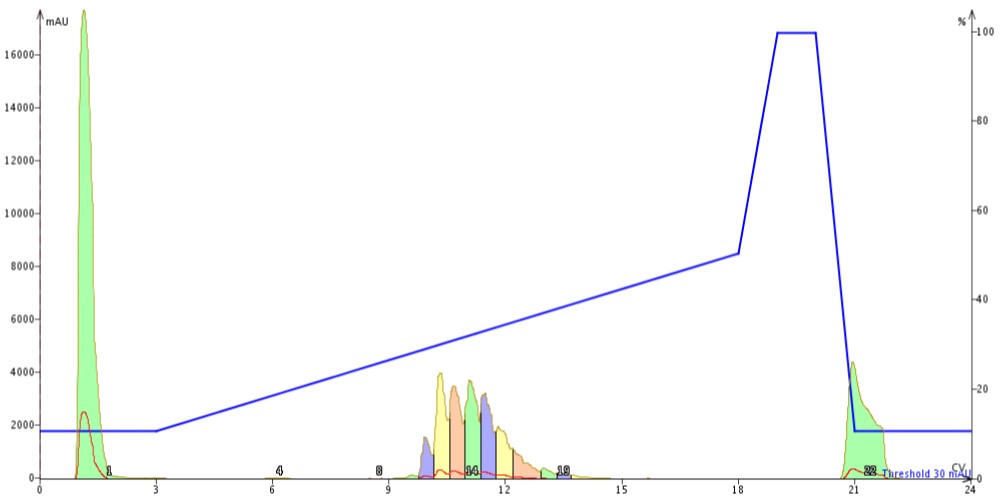 Figure 3: Results of revised mobile phase gradient in which acetonitrile concentration increases from 10% to 50% over 15 column volumes.
Figure 3: Results of revised mobile phase gradient in which acetonitrile concentration increases from 10% to 50% over 15 column volumes.
As long as I am experimenting with a peptide that doesn’t really matter to me anyway, let’s take a page from the organic flash purification strategy book and try a step gradient. This method, commonly used during small molecule purifications, utilizes a series of isocratic holds to improve separations in fewer column volumes than would be necessary for a traditional linear gradient and but not often in peptide purifications. I used the optimize button in the software to help me define the acetonitrile concentrations at which to insert the isocratic holds, injected slightly more peptide sample and pushed play! The separation does not seem to improve for this particular sample, although I was able to completely resolve a small peak with the second gradient step, Figure 4. This may be an interesting strategy for extracting a more pure peptide sample from a crude mixture that I will work on in the future.
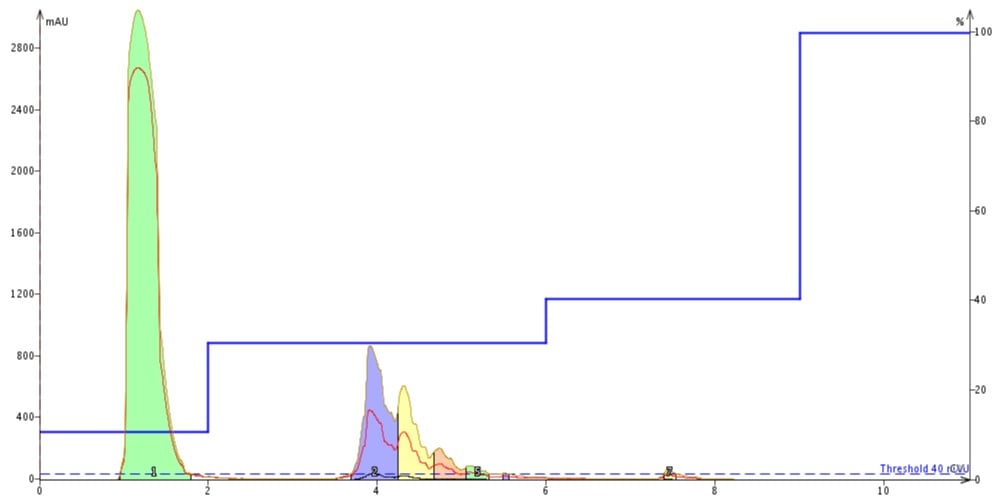 Figure 4: Results of step gradient purification.
Figure 4: Results of step gradient purification.
All in all, this was a fun little exercise in purification! The software was extremely easy to learn and subsequently expand upon to modify my methods however I desired. The purification could have been better, but I anticipate this will also improve when I handle my peptides properly. Even if the chromatography didn't improve, I am able to load almost an entire synthesis onto a single cartridge, achieve a modest level of purity very quickly, leaving me with only a few injections to completely purify my sample by HPLC. Most importantly, the chromatography exercise I described above took approximately 90 minutes of real time, a fraction of the 3 and a half hours it could have taken to optimize the HPLC gradient for a completely unknown peptide. In the future, I will continue to explore the utility of flash chromatography to purify my peptides! If nothing else than to speed up the purification process. For those samples that demand very high purity, I will certainly utilize HPLC for final purification steps (with hopefully fewer injections this time around).
To learn more about Biotage's solutions for peptides purification using high performance flash chromatography, click the link below.
Published: Jan 30, 2023 5:39:31 PM

