After my seminars, I often receive questions from astute attendees on the topic of activated carbon, and how the more modern powerful approach of metal scavenging compares to the traditional application of activated carbon. This can be an emotive topic, sometimes the question can be a blunt as ‘What’s wrong with carbon? It’s the way most process groups have historically removed transition metals such as palladium.'
So, it’s a fantastic question and one that we looked into at Biotage in some dedicated small studies. We took a model Active Pharmaceutical Ingredient (API), spiked it with a known amount of palladium and looked at the efficiency of various mitigation processes typically used to remove the palladium.
We investigated the use and application of solid supported metal scavengers as compared with activated carbon in this case. Both media are completely insoluble adsorbents that may be added to reactions, which can bind metal, and can be removed ‘cleanly’ afterwards, perfect heterogeneity. But as we found out, not all metal scavengers are the same….
We initially focused our attention on three powerful industry established scavengers for palladium, Biotage® MP-TMT, ISOLUTE® Si-TMT and ISOLUTE® Si-Thiol, Figure 1.
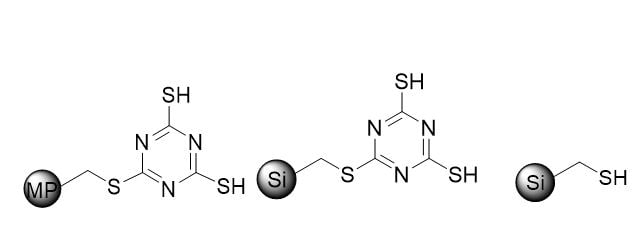
Figure 1. structures of solid-bound scavenger media. Left – Biotage® MP-TMT. Middle – ISOLUTE® Si-TMT, Right – ISOLUTE ® Si-Thiol.
The style of the experiment was simple, we added a defined amount of metal scavenger to a reaction volume and simply stirred / filtered to work up and process. Datapoints were taken, which allowed us to correlate the amount of scavenger added vs resulting palladium by elemental analysis and the results plotted below, Figure 2.
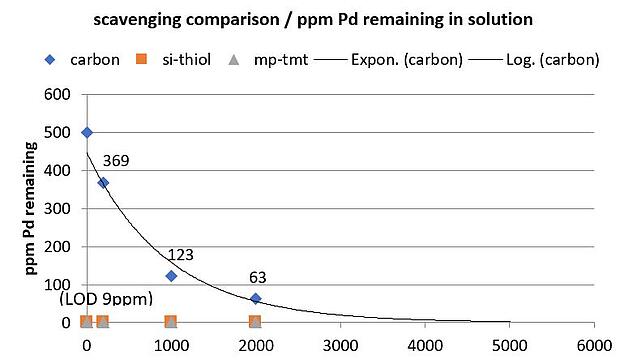
Figure 2.Mass of scavenger added (mg) vs the resulting Pd in solution (ppm), starting from an organic 500 ppm Pd solution.
Under these conditions, activated carbon removed some metal from solution but failed to remove 100%, even at the experimental maximum load of 2 g/10 mL, almost 13% Pd remained after 16 hours. Activated carbon has a bulk density of approx. 2.9 mL/g (0.34 g/mL), thus our carbon treatment consumed almost 3 mL of reactor vial space (total solvent volume 10 mL). By extrapolation, 5 g (14.5 mL) of activated carbon would have been necessary to remove most of the metal, and put simply, if implemented, there was not going to be enough space in the reactor vessel if we continued on this route.
In contrast, the metal scavengers were much more effective, removing 98.2%+ of the Pd, even at the lowest load of 0.2 mg/10 mL. Bulk density of the metal scavenger is ~1.66 mL/g (0.6 g/mL), suggesting that for the lowest load, only 0.3 mL of reactor space was required.
What did surprise me though, and to complete the picture, was the story that unfolded when we looked at chemical yield and recoveries.
At the experimental maximum load of carbon (2 g), we achieved 88% palladium reduction, but recovered only 46% of our API analogue. The 88% reduction may sound good, but in reality, this is a very poor result. Removing 88% of the 800 ppm Pd remaining after a 0.5-1% catalytic Suzuki reaction, is a clear failure by most metrics of metal removal and will necessitate a repeat treatment or re-work (that’s still ~100 ppm left). Twenty years ago maybe it would have been fine but not really in today’s standards and compliance orientated environment. In comparison, the same treatment using a smaller amount of Biotage® Si-Thiol resulted in >98.2% removal, and 100% (quantitative) recoveries.
So, coming back to the initially posed question, ‘Are metal scavengers better than carbon’…well, carbon works, but there may be less rationale for using it as a generic adsorbent. Carbon may not always remove palladium as effectively, definitely consumes more reactor space (thus could be more difficult to clean afterwards), and also risks reducing precious yield recoveries of our valuable API /product.
In our examples, carbon was able to scavenge palladium but the amount of carbon required to completely remove palladium would have had a devasting effect on reaction yield, while also barely fitting in the reactor in the first place. This would have further detrimental effects to mass transport, solvent usage, reaction kinetics , and additional solvent waste implications.
Read our previous blog post to learn more, just follow the link below!

